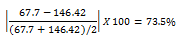Vitamin C Analysis
What is Spectrophotometry:
|
Spectrophotometry was also discussed on the Protein Analysis page. Below will detail a short recap of the process:
Spectrophotometry: a process that measures the absorbance of light from a sample. A spectrophotometer is a machine that shines a light beam through a sample and the detector in the machine measures the intensity of the light, to provide an absorbance reading. The spectrophotometer can be used to analyze many different types of samples, such as protein, as indicated on the Protein Analysis page, as well as ascorbic acid concentration in a food, as performed in this experiment. |
Additional Information: |
For more information regarding spectrophotometry, please go here
|
Purpose of Experiment:
|
To determine the concentration of ascorbic acid in the fruit juice sample (orange juice) by using the spectrophotometer
|
Procedures:
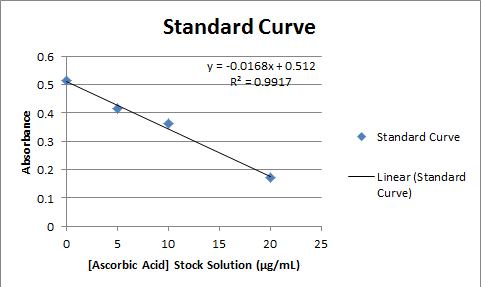
Standard solutions were made in 0, 5, 10, and 20 micrograms/ml concentrations by using the 200 micrograms/ml ascorbic acid stock solution. The following table shows the dilutions made:
Two mL of each of these 4 dilutions were added to a test tube, with 3 mL of sodium citrate buffer, and covered with parafilm.
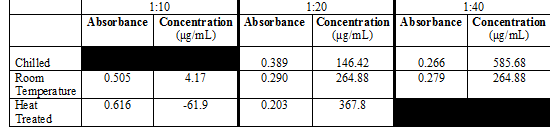
Then the dilutions of the chilled juice, room temperature juice, and heated juice samples were prepared, in duplicate. The chilled orange juice was diluted 1:20 and 1:40 with citrate buffer. The room temperature orange juice (the control group) was diluted 1:10, 1:20, and 1:40 with citrate buffer. The heated orange juice was diluted 1:10 and 1:20 with citrate buffer.
Once these dilutions were made, a blank that consisted of only the sodium citrate buffer was used to blank the spectrophotometer. Then to calculate the absorbance of each of the dilutions, 1 mL of DCIP was added to 1 mL of each of the standard dilutions into a cuvette, and 0.2 mL of DCIP was added to 1 mL of each of the juice samples into a cuvette. The absorbance values were read for each sample
Note: add the DCIP one tube at a time, NOT all at once because the color is stable only for 5 minutes.
Then the dilutions of the chilled juice, room temperature juice, and heated juice samples were prepared, in duplicate. The chilled orange juice was diluted 1:20 and 1:40 with citrate buffer. The room temperature orange juice (the control group) was diluted 1:10, 1:20, and 1:40 with citrate buffer. The heated orange juice was diluted 1:10 and 1:20 with citrate buffer.
Once these dilutions were made, a blank that consisted of only the sodium citrate buffer was used to blank the spectrophotometer. Then to calculate the absorbance of each of the dilutions, 1 mL of DCIP was added to 1 mL of each of the standard dilutions into a cuvette, and 0.2 mL of DCIP was added to 1 mL of each of the juice samples into a cuvette. The absorbance values were read for each sample
Note: add the DCIP one tube at a time, NOT all at once because the color is stable only for 5 minutes.
Results:
Ascorbic Acid Concentration in the Juices
Discussion/ Improvements:
Since the data points are more accurate in the middle of the standard curve, the focus will be placed on the 1:20 dilutions, which have an absorbance close to 0.300. When looking at these three samples, the heat treated sample appears to have the highest ascorbic acid concentration, with a concentration of 367.8 μg/mL Then the room temperature sample appears to have the second highest ascorbic acid concentration, with a concentration of 264.88 μg/mL, and the chilled sample has the lowest ascorbic acid concentration, with a concentration of 146.42 μg/mL
Typically, vitamin C is supposed to degrade when heated, which indicates that the ascorbic acid concentration should be lower for the heat treated orange juice. However, this was not observed in this experiment. A possible explanation of these wide ranging results is that the three juices were sitting at room temperature for approximately 2 hours prior to the use of the spectrophotometer to determine their absorbance. This may have altered the difference between “chilled,” “room temperature,” and “heat treated,” which influenced the ascorbic acid content in the different samples.
The concentration listed on the carton was 160% Vitamin C per the Daily Value, which is 60 mg, and converts to approximately 96 mg. Since 355 mL of concentrate and 1062 mL of water was used to form this juice, this would be 96 mg/1417 mL of orange juice. In order to compare this value with the actual value obtained from the experiment, the conversion from mg/mL to μg/mL occurred, as shown below:
Typically, vitamin C is supposed to degrade when heated, which indicates that the ascorbic acid concentration should be lower for the heat treated orange juice. However, this was not observed in this experiment. A possible explanation of these wide ranging results is that the three juices were sitting at room temperature for approximately 2 hours prior to the use of the spectrophotometer to determine their absorbance. This may have altered the difference between “chilled,” “room temperature,” and “heat treated,” which influenced the ascorbic acid content in the different samples.
The concentration listed on the carton was 160% Vitamin C per the Daily Value, which is 60 mg, and converts to approximately 96 mg. Since 355 mL of concentrate and 1062 mL of water was used to form this juice, this would be 96 mg/1417 mL of orange juice. In order to compare this value with the actual value obtained from the experiment, the conversion from mg/mL to μg/mL occurred, as shown below:
The highest absorbance reading obtained from the 1:20 dilution samples was the chilled orange juice sample, with an absorbance of 0.266. The ascorbic acid concentration calculated was 146.42 μg/mL. This value greatly differs from the expected value of 67.7 μg/mL, with a percent difference of 73.5%.