Titratable Acidity
What is titratable acidity?
|
The measure of ionized and unionized forms of the acid present in the sample
|
Purpose of Experiment:
|
To determine the acidity of three unknown acids, using a pH meter to measure the pH of the solution and performing a titration to obtain the titratable acidity
|
Procedure:
Pour 50 mL of acid samples A, B, and C into separate beakers.
Add five drops of phenolphthalein and methyl red.
Calibrate pH meter using the three buffer solutions.
Clean the 25 mL buret using .2M NaOH.
Fill the buret with .2M NaOH.
Place one beaker containing an acid on magnetic stirrer.
Gently stir and titrate the acid samples using 1 to 10 mL increment.
Note the color change and pH at each increment.
Add five drops of phenolphthalein and methyl red.
Calibrate pH meter using the three buffer solutions.
Clean the 25 mL buret using .2M NaOH.
Fill the buret with .2M NaOH.
Place one beaker containing an acid on magnetic stirrer.
Gently stir and titrate the acid samples using 1 to 10 mL increment.
Note the color change and pH at each increment.
Results:
|
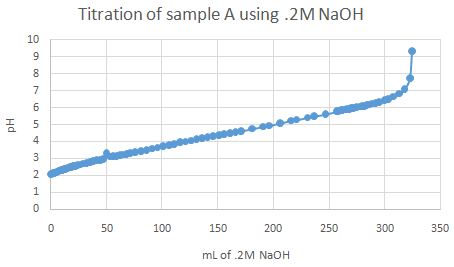
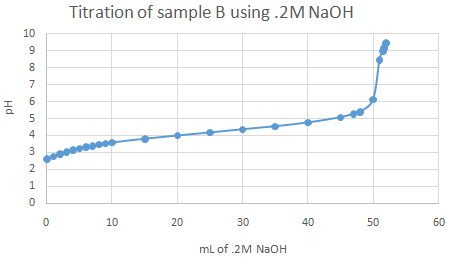
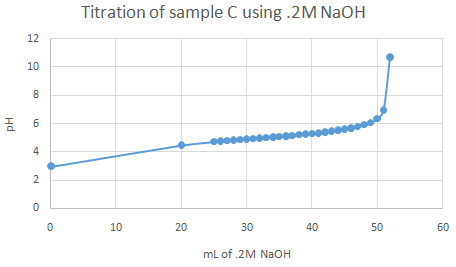
The important pka values from the graphs (half equivalence points) are as follow:
For acid A, pka is 5.97 (turned red from fuchsia). For acid B, 4.56 to 4.78 (turned peach from fuchsia). For acid C, 5.51 (turned clear orange from fuchsia). Identifications are - A = ascorbic acid, B = citric acid, C = acetic acid Observations:
Theoretically, more than one point of inflection should have been spotted upon graphing, but only one half equivalence point (or range) was found for each acid sample.
Calculations:
Titratable acidity for sample A: 176.12g/mol (ascorbic acid), .1M NaOH * .325L NaOH = .0325 mol
Acid and NaOH react 1:1 ratio. .0325 mol * 176.12 g/mol = 5.724 g % Acidity for sample A = (.1N * 325mL * 88.06)/(100mL * 10) = 2.86 % acid Titratable acidity for sample B: 192.124g/mol (citric acid), .1M NaOH * .520L NaOH = .0520 mol Acid and NaOH react 1:3 ratio. Thus .0520/3 = .0173 mol .0173 mol * (100mL/50mL) * 192.124 g/mol = 6.647 g % Acidity for sample B = (.1N * 520mL * 64.04)/(100mL * 10) = 3.33 % acid Titratable acidity for sample C: 60.05g/mol (acetic acid), .1M NaOH * .052L NaOH = .0052 mol Acid and NaOH react 1:1 ratio. .0052 mol * (100mL/50mL) * 60.05g/mol = .625 g % Acidity for sample C = (.1N * 520mL * 60.05)/(100mL * 10) = 3.12 % acid |
|
Discussion/ Improvements:
Although we carefully titrated with minute increments, we had to gradually increase the increment to as much as 10 mL, since pH or color change were negligible. If our group were to complete this experiment again, I would set up 50mL buret (filled with NaOH) on both sides of the clamps, then titrate same acid samples simultaneously on each beaker. If pH meter is not available, replication of titration of all the acid samples would yield more accurate results. Also, it was quite frustrating how there were not enough inflection points to extract and compare pKa values. For sample A especially, the amount of .2M NaOH used was too much. As learned from the lecture on Wed, Feb 11th, 2015, titratable acidity includes other flavors, which is different from acidity. Active acidity is H+, and pH, which deters microbial growth.