Sodium in Sports Drinks
What is Mohr Titration?
|
Mohr Titration is a direct titration method to quantify cholride ions in a solution. The direct titration of chloride ions will allow us to indirectly quantify the sodium ions in a solution. Mohr Titration is one of many precipitation titration methods. In Mohr Titration, a solution containing chloride ions and potassium chromate is titrated with silver nitrate. Silver nitrate dissolves in solution and the free silver ions will complex with the free chloride ions. Silver ions will complex with free chloride ions until all of the free chloride ions are taken up, leaving no free chloride ion. Now, any excess silver nitrate added will complex with the chromate that was added to the solution. When silver and chromate complex together, an orange colored solid called silver chromate is formed. The formation of the orange color signals the end of the titration. The volume of silver nitrate needed to complex all the free chloride ions has been reached when the solution turns orange. This volume is noted and can be used to calculate the amount of chloride ions in solution. To the left is a picture of the experimental set up!
|
Purpose of Experiment:
|
To use the Mohr Titration method to determine the sodium content of a sports drink sample (Gatorade Frost-Glacier Cherry). Click here to read about why sodium in sports drinks is important!
|
Procedure:
Before beginning, we had to standardize the silver nitrate we were going to use to titrate the Gatorade sample. Standardizing simply means simulating a titration trial with a solution that has a known amount of chloride ions. We added a known amount of potassium chloride to water and added potassium chromate into the solution. Silver nitrate was used to titrate the sample and the volume needed to completely reach the orange color was noted. With this volume, we were able to calculate the molarity of the silver nitrate solution we were using in the experimental trials. Next, we took 5mL of Gatorade sample and mixed it with boiling water. With this solution, we were able to run duplicate titration trials with our standardized silver nitrate.
Results:
The following are the results from the standardization of silver nitrate:
When we titrated the standard solution we first observed a yellow color because of the addition of the potassium chromate indicator. Adding the silver nitrate turned the yellow into an orange-pink color. The endpoint was observed when the orange-pink color persisted.
For the first duplicate of our standard sample, we did not precisely add silver nitrate. This first run allowed us to get a rough estimate of when we should expect the color change. Our second duplicate was titrated precisely, with 0.1mL increments as we got close to the expected endpoint:
When we titrated the standard solution we first observed a yellow color because of the addition of the potassium chromate indicator. Adding the silver nitrate turned the yellow into an orange-pink color. The endpoint was observed when the orange-pink color persisted.
For the first duplicate of our standard sample, we did not precisely add silver nitrate. This first run allowed us to get a rough estimate of when we should expect the color change. Our second duplicate was titrated precisely, with 0.1mL increments as we got close to the expected endpoint:
Following the same titration process, we now used a Gatorade sample. We overshot the endpoint for the first trial because of the small quantity needed, but we were able to accurately get an endpoint for our second trial:
Our dilution factor is 100 because we added 5mL of Gatorade to 95mL of boiling water. We will use this factor in the following calculations:
Standardization of 0.1 M silver nitrate:
We calculated our silver nitrate molarity from Standard 2 and we got a value of 0.1003 M. We expected a value around 0.1 M.
Sodium Determination in Gatorade:
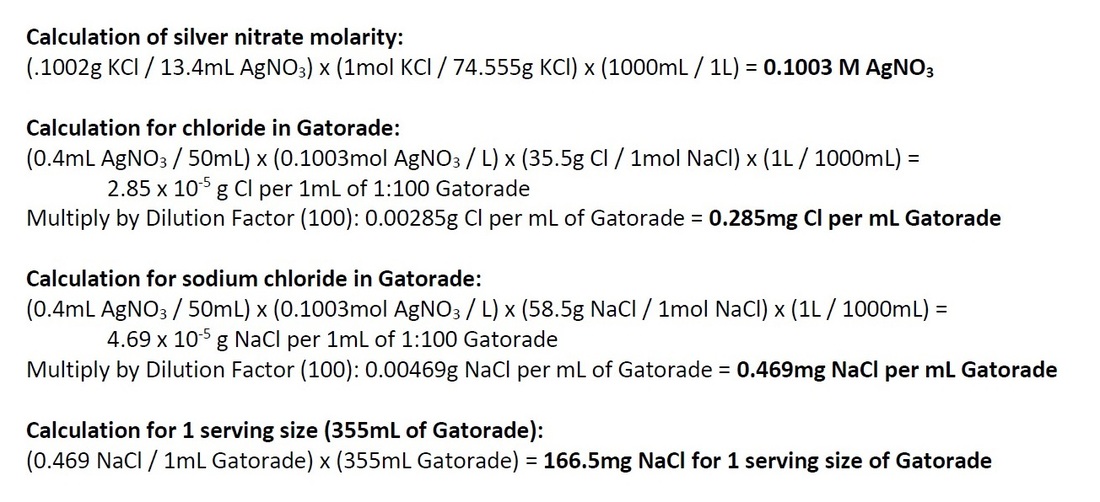
We calculated sodium content from Standard 2 and we got a value of 0.469mg NaCl per mL Gatorade.
Below are the calculations:
Standardization of 0.1 M silver nitrate:
We calculated our silver nitrate molarity from Standard 2 and we got a value of 0.1003 M. We expected a value around 0.1 M.
Sodium Determination in Gatorade:
We calculated sodium content from Standard 2 and we got a value of 0.469mg NaCl per mL Gatorade.
Below are the calculations:
Discussion/ Improvements:
The nutrition label on the Gatorade bottle says that there is 160mg of sodium for a 355mL serving size. We calculated that there is 166.5mg NaCl for one serving size of Gatorade. This comes out to a percent error of about 4%. The values were close, but more precision in titrating may get us closer to the true value on the nutrition label. With regards to the % daily value, the recommended intake of sodium is 2400mg. 160mg of the 2400mg on the nutrition label is 6.7% (rounded to 7% on the label). If we put 166.5mg of sodium over the 2400mg recommended, we get 6.9% of our daily value. This value is also close to the 7% on the nutrition label.