Lyophilization (Freeze Drying)
What is Freeze Drying?
|
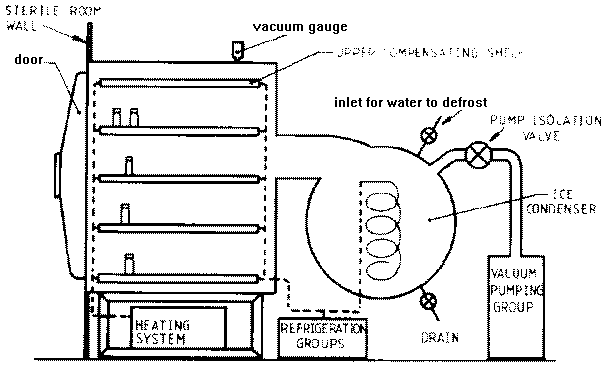
Freeze drying is a process employed to extract all water from a particular food matrix without exposing the sample to the atmosphere. After dropping to -40 degrees C, well below the triple point for water, a heat source located below the main chamber of a freeze dryer raises the temperature just high enough that water sublimates from the food matrix, beginning with the external particles and moving towards the center. The water vapor is then pulled into a secondary chamber by a vacuum, and undergoes deposition once it comes in contact with a -100 degree C coil, transitioning directly from gas to solid form.
|
Purpose of Experiment:
To extract water quickly from a chosen food matrix without exposing the sample to the atmosphere or any external source of heat.
Procedure:
The weight of a single plastic bag was taken, then used to contain one serving size of our food product, canned Chef Boyardee Lasagna. Once the bag was labeled with section number and group identification, it was placed into a freeze drying chamber with an internal temperature below the freezing point of water that transitioned the water content of the food sample directly from a solid to a vapor (sublimation), allowing for the extraction of all water from the food matrix without exposure to heat. Water molecules closest to the edge of the sample migrated out first, and so forth until all the water was removed.
Results:
- Weight of one serving size prior to lyophilization: 258.2 g
- Weight following lyophilization: 58.38 g
- Net loss (water content of one serving size of food product): 258.2 g-58.38 g = 199.82 g
- % water: 199.82g/258.2g*100= 77.39% water
Discussion/ Improvements:
- Our sample had a very high water content, with less than 25% of the original mass remaining after the freeze-drying process.
- Improvements:
- Increase the surface area more to ensure maximal water loss.